We often find neon used in signs as it is coloured when electricity is put through it. These gases are quite special, when a high amount of electric energy is put through them they will give off a coloured light. The Halogens are next to group 18, the noble gases these are non metal gases that are very unreactive and are all gases. A common gas in group 17 is chlorine which is used to kill bacteria and clean swimming pools, it has a strong smell. From the position of the diagonal line in the periodic table separating metals and nonmetals, classify the group 13 elements. A larger atom in the non-metals produces stronger attractions meaning the states change from gas to liquid to solid progressing down Group 17. Progressing from Fluorine to Chlorine both gases to Bromine, a liquid to Iodine a solid. Group 17 the Halogens contains a range of non-metals but a key trend can be seen. Nitrogen is also found in the air and is important for plants an animals to grow. Between the metals and the non-metals lie the elements which. 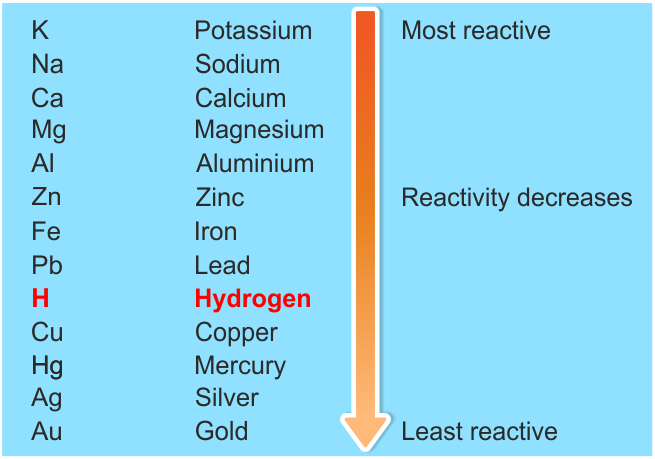 
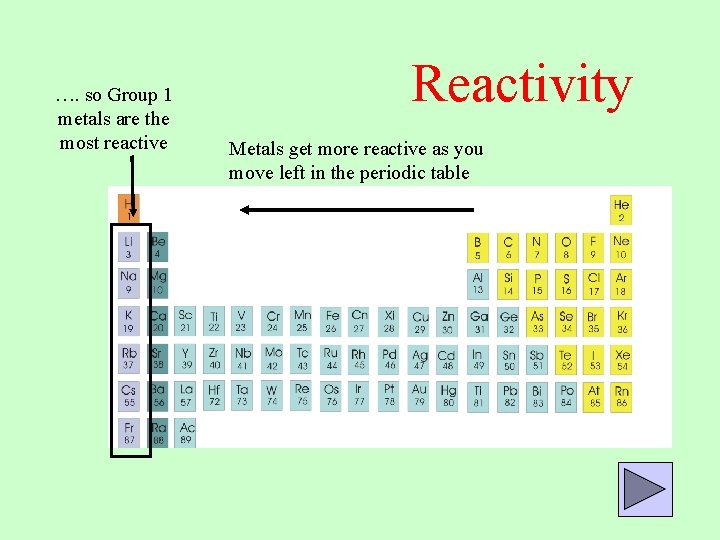
Metals occur on the left-hand side of the Periodic Table and non-metals on the right-hand side. Carbon is found in a lot of living organisms as a compound with other elements, it is also used as coal in power stations. This trend occurs due to atoms more readily accepting electrons to fill their valence shells rather than losing them to have the previous, already full, electron shell as their outer shell. Oxygen is in the air and is important to us when we breathe and for respiration. Some elements in these groups you will be familiar with are oxygen, nitrogen and carbon. Metals also tend to be shiny and silver coloured where as non metals tend to be coloured and sometimes dull. Non metals in this part of the periodic table tend to be liquids and gases and are brittle, when they are hit they break apart and shatter. These non metals have very different properties to metals, they do not tend to barde shiny and wont conduct heat or electricity well. For example Group 15 contains the non-metals Nitrogen and Phosphorus, the semi-metal Arsenic and the metals Antimony and Bismuth. They are often used in buildings and for making strong materials as they are heavy and hard.įrom Group 13 to 16 these are a mixture of metals, non-metals, and semimetals. These have characteristic properties and are less reactive. The next part of the periodic table is the transition metals from group 3 to 12. They also have similar reactivity with Group 1 being much more reactive than Group 2. Group 1 alkali metals, Group 2 Alkaline earth metals are both named as such as often they form alkalis. Metals are predominantly located on the left of the periodic table. There are of course some exceptions to this. Most metals are solids and non-metals are liquids and gases.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
